gczop
Full Member
  
Posts: 202
|
Post by gczop on Mar 2, 2016 16:38:33 GMT -6
Hey Gordon, I didn't realize Lithium Carb wouldn't decompose, I assumed that being a strong flux it would have. I've considered not using it because it's soluble, but it does allow for certain compositions that I don't think can be met with other lithium sources. Also, I don't recall hearing of Lithium Oxide as a glaze material. Do you use it, and if so where do you get it? Hi Evan, Its a minor comment, something to be aware of. The carbonate will decompose freely approaching cone 10. This eutectic melts at about 400c: (I've tried it) Carbonate eutectic mixture (32.1 wt% Li2CO3; 34.5 wt% K2CO3; 33.4 wt% Na2CO3) . |
|
|
|
Post by jfox on Mar 2, 2016 20:21:40 GMT -6
|
|
|
|
Post by evan cornish-keefe on May 14, 2016 10:48:29 GMT -6
|
|
|
|
Post by tileman2 on May 15, 2016 20:00:10 GMT -6
Evan:
I know you are still zoning in; but I like the iridescent look that it has now. I have not fired much since last December: been working primarily on clay bodies. Looks like it will be awhile longer, I bought a house at an estate sale to flip.
Tom
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on May 18, 2016 17:51:06 GMT -6
Evan,
Perhaps the glaze is a bit quirky, haven't see anything like what you are getting.
Maybe try a firing with two pieces, one with moly and one without. To paraphrase Yogi Berra, you can observe a lot by looking.
Gordon
|
|
|
|
Post by evan cornish-keefe on Jun 19, 2016 23:30:13 GMT -6
Hey Tom, I've been making some progress with a very translucent cone 6 clay, soon ill get back into testing Mo glazes on that clay.
Hey Gordon, yes quirky is a polite way to put it. That's a great Yogi Berra quote, I also like: When you come to a fork in the road, take it.
|
|
|
|
Post by hollymckeen on Jul 5, 2016 18:03:28 GMT -6
I posted some moly pics of new tests over on the everything else thread.
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Aug 27, 2016 9:12:48 GMT -6
Moly tests   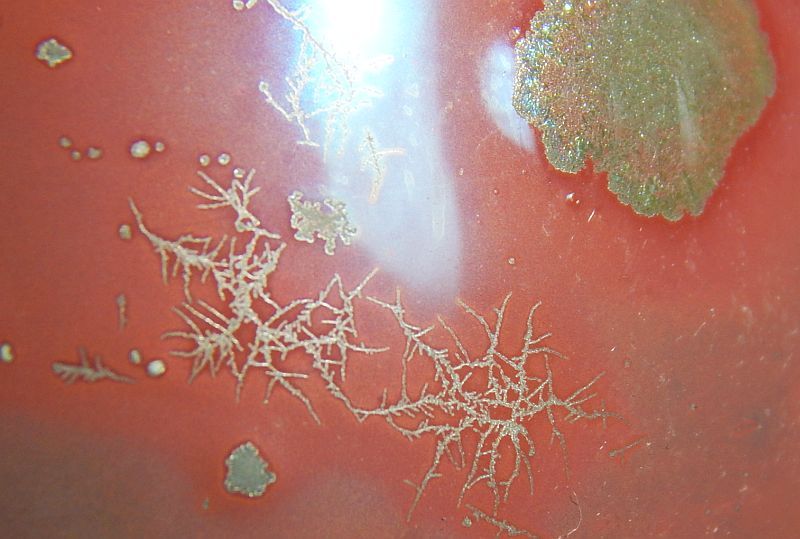 This a dish with MnO2 and TiO2 in the bottom and custer feldspar glaze with iron oxide on the side. Some type of crystal appears to have grown from the bottom.  |
|
|
|
Post by John Garrison on Oct 11, 2016 20:21:09 GMT -6
|
|
annie
Junior Member
 
Posts: 55
|
Post by annie on Oct 17, 2016 8:16:42 GMT -6
 Yippee. Under close inspection, I found I have a couple of the black crystals like John.  Here's the piece this was found on (lower right corner in from the rim, about 5 0'clock). 3110 glaze with Co and Mn02; no Molybdenum. Also has some titanium and alumina hydrate. Could they be from either of those?   --Annie |
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Oct 22, 2016 8:25:33 GMT -6
Hi Annie, This in a 3110 base with 2% each of Manganese, Cobalt and Titania. Evan Cornish Keefe has gotten some nice ones. Thanks for your Moly comment.  These, below, in a Baria, Titania and Silica mix about the same proportions as Benitoite, the California State Gem. Cheers, Gordon  |
|
annie
Junior Member
 
Posts: 55
|
Post by annie on Oct 23, 2016 1:35:48 GMT -6
Really beautiful Gordon! So we know we both have same materials in our glazes. I noticed many more of them on pieces in a different clay body. Maybe mix from the clay and glaze materials. Bella!
|
|
bbucky
New Member
 Married
Married
Posts: 36
|
Post by bbucky on Oct 24, 2016 1:02:31 GMT -6
How did you get pictures to post?
Bbucky
|
|
annie
Junior Member
 
Posts: 55
|
Post by annie on Oct 25, 2016 1:19:31 GMT -6
Greg helped me. I used Google+ free photo app to load the pics for a URL. Had to move them to a "shared" folder in Google's photo app. Once in the shared photo folded, I could right click to select copy image location open to public now. Then when posting my comments on this forum, I selected the insert image icon on the tool bar, looks like a landscaped pic) then pasted the copied link/address to the first box (pasted right on top of the "http://" text.
I just learned how myself, with help. And it has been 50/50. To test I logged out of google+ and then selected "preview" in my post. Before when not in the Google share folder I saw them but no one else did.
Greg has tutorial in the forum. Also, I tried Photobucket before but because of all the ads it was hopelessly slow. Google worked better for me once Greg (Mowhawk Piper) explained about the share folder.
-Annie
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Feb 26, 2017 10:05:00 GMT -6
Perovskite?!  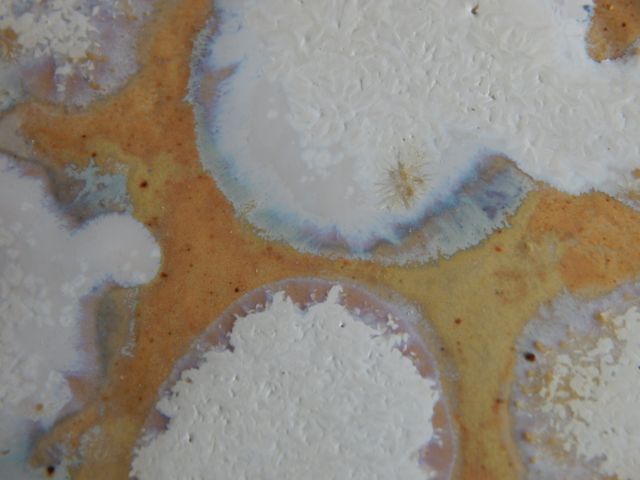 Photo 1 one part each of whiting, titania and 413 frit cone ten. refired with left half covered with 413 base (68% 413, 24% ZnO and silica). Photo 2 one part whiting one part titania and two parts 413 frit, portions of 4 globs shown. Left and top globs covered with 413 base, other two not. Top glob has Mn compound added to the 413 base(note crystal similar to my avatar). Single firing cone 10. Cheers, Gordon |
|