|
|
Post by tileman2 on Nov 8, 2015 18:35:08 GMT -6
Kuba:
Love the close up pics. The second one would make for a nice poster: reminds me of ice crystals forming. Will give me something to study, I see all kinds of information in them.
Tom
|
|
|
|
Post by evan cornish-keefe on Nov 9, 2015 12:19:08 GMT -6
Thanks Gordon! Now I'm looking I see the snowflakes quite often, just large enough to see without a microscope. Here's some in the bottom of a bowl, cone 5 with 3% each RIO and MnO2. Close up taken with my cell phone.   Kuba, great colors you're getting! |
|
paul
Junior Member
 
Posts: 51
|
Post by paul on Nov 10, 2015 16:19:32 GMT -6
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Nov 17, 2015 8:50:08 GMT -6
Astrophyllite analogue  |
|
Kuba
Full Member
   SztukKilka in Old Formu
SztukKilka in Old Formu
Posts: 111
|
Post by Kuba on Nov 17, 2015 13:42:24 GMT -6
WOW!
|
|
|
|
Post by evan cornish-keefe on Nov 24, 2015 22:05:42 GMT -6
Nice Gordon! Is that with manganese or iron? I decided to try a USB microscope, $25 on eBay. It'll zoom up to 200X, which seems more than enough for this, the camera is 5 megapixels and the image is very blurry. It's a joke of a microscope, but all the other photos i took were with an iPhone (8 megapixels) held up to the eyepiece.   |
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Nov 27, 2015 9:23:17 GMT -6
Evan, Here's another flower, FOV about 2". (BTW Mn used in the astro glaze.)  |
|
|
|
Post by evan cornish-keefe on Dec 3, 2015 13:06:53 GMT -6
Awesome Gordon! I'm curious what your firing schedule looks like, and what effect that has on the size and shape of these crystals? Are the rough looking edges from the way the kiln is cooled? The Mo crystals Im getting are mostly tiny, and the shapes seem very random, though i haven't tried different cooling cycles much yet.       |
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Dec 4, 2015 12:12:29 GMT -6
Evan,
its my conjecture that subtle changes in the pertinent parameters affect the crystal morphology. Here we fire from 120c to cone ten in five hours to save on electric. The firing schedule isn't changed but the result varies. As your work and Joerg's dendrites show, there is a remarkable range of crystal appearance possible.
|
|
|
|
Post by evan cornish-keefe on Dec 7, 2015 11:11:00 GMT -6
So you don't slow cool? Is the difference in your results just the size & shape of the crystals, or more significant?
Do you think it's more to do with glaze chemistry than the firing?
|
|
|
|
|
|
Post by evan cornish-keefe on Feb 14, 2016 10:02:34 GMT -6
Dang Gordon, you have some great things going! It would be great to get some thoughts on the last Mo tests I tried. willemite.tumblr.com/post/134732115590/powellite-testsI assume the small white spots are Powellite, the florescence looks the right color and I get more as the amount of calcium in the glaze increases. So far the iridescent interesting shaped crystals don't seem to form without some Titanium in the glaze. Difficult to see in these blurry photos, but Mo in this Fe Ti crystal glaze creates colorful bands around metallic bronze colored crystals. 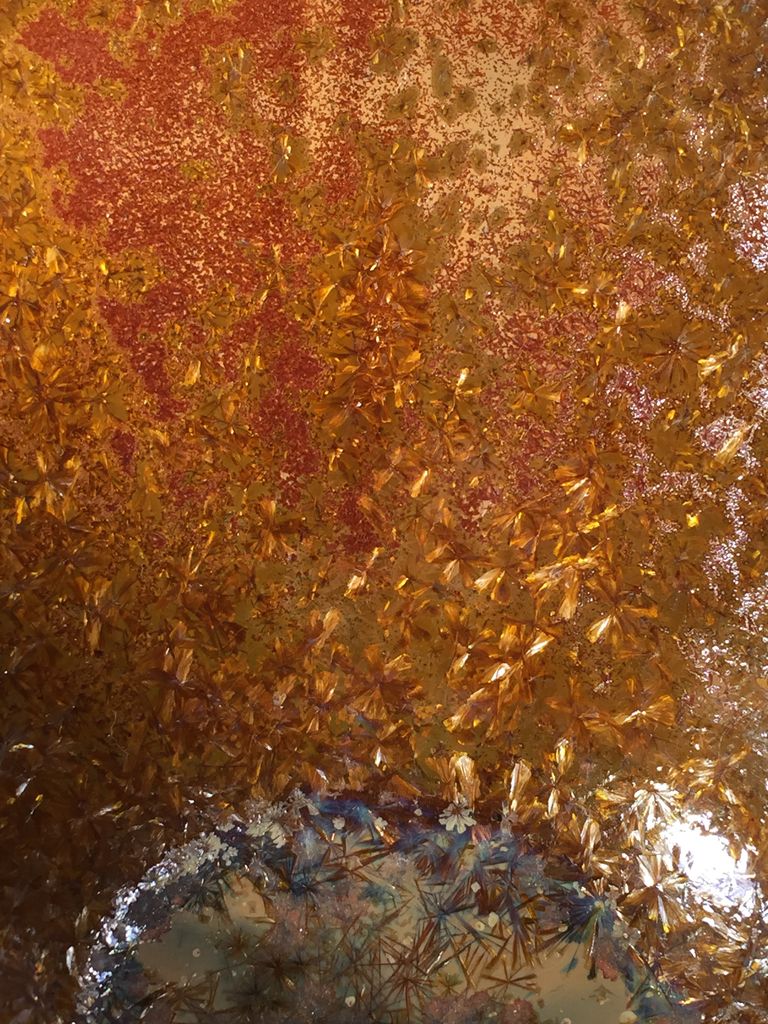    |
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Feb 14, 2016 17:22:56 GMT -6
Hi Evan,
The crystals you are getting are fantastic.
Lamping your results with UV is an effective diagnostic tool.
You are on the right track with that series of tests.
I have a bias against using Lithium Carbonate as a flux at the temperature you are firing.
Calcium carbonate decomposes freely to CaO and CO2 just under 1800F, for example, while Li2OCO2 doesn't do so until approaching 2400F.
The question being, is Lithium Carbonate behaving the same way as Lithium Oxide (in a glaze)?
Simpler is better, at least here. Gordon
|
|
|
|
Post by evan cornish-keefe on Feb 28, 2016 18:48:09 GMT -6
Hey Gordon,
I didn't realize Lithium Carb wouldn't decompose, I assumed that being a strong flux it would have.
I've considered not using it because it's soluble, but it does allow for certain compositions that I don't think can be met with other lithium sources.
Also, I don't recall hearing of Lithium Oxide as a glaze material.
Do you use it, and if so where do you get it?
|
|
am
New Member

Posts: 3
|
Post by am on Feb 29, 2016 17:56:54 GMT -6
|
|