|
|
Post by evan cornish-keefe on Jul 22, 2015 20:53:50 GMT -6
Tom, are these the images you're referring to? 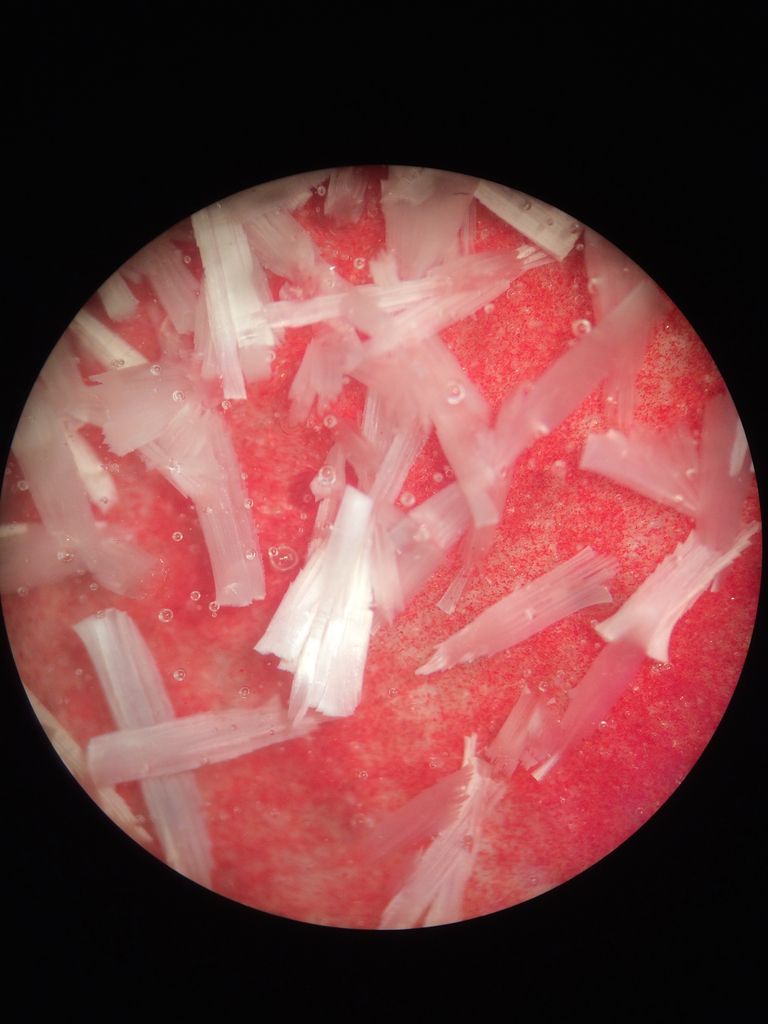 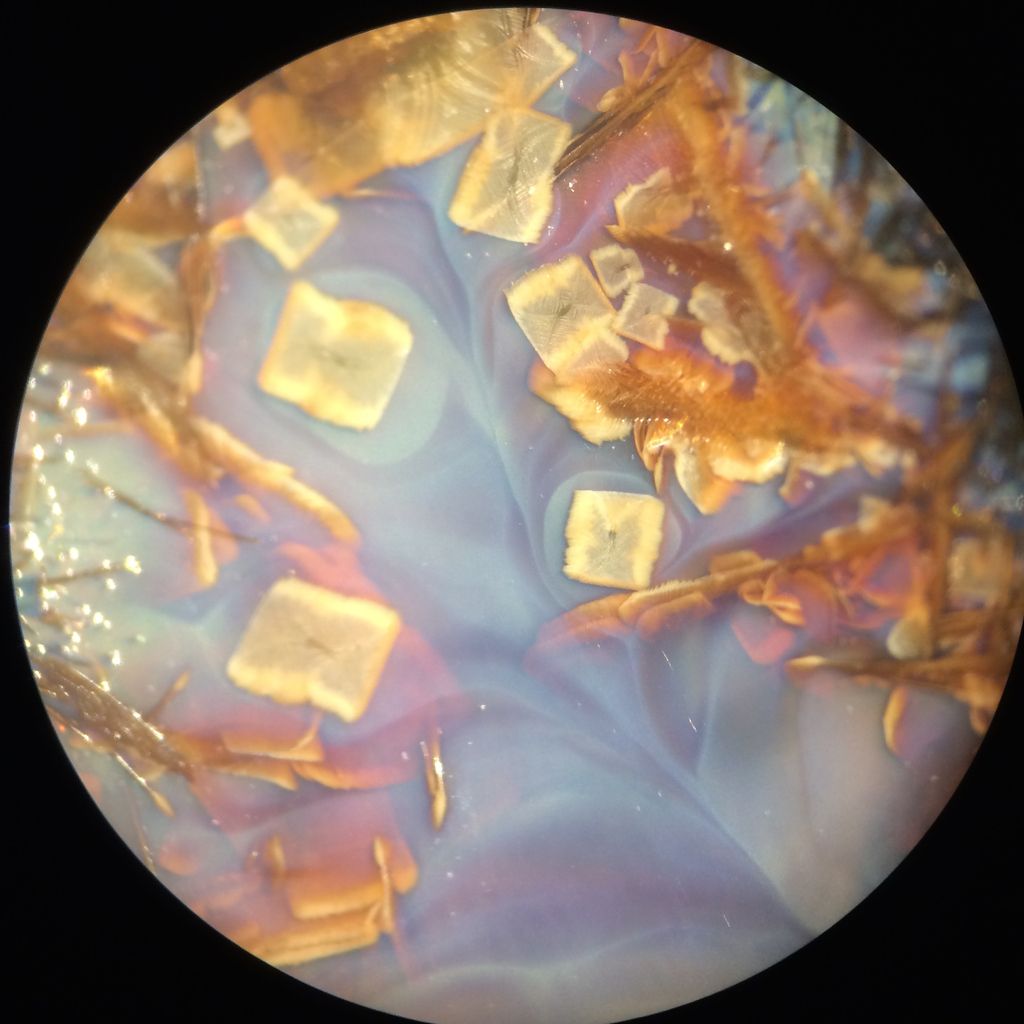 Kuba, if no manganese is in yours I have no clue, still I might try swapping Manganese for Zinc. Here is a Willemite crystal glaze with about 5% MnO2, it usually doesn't have these secondaries, and I don't know why they formed this time. 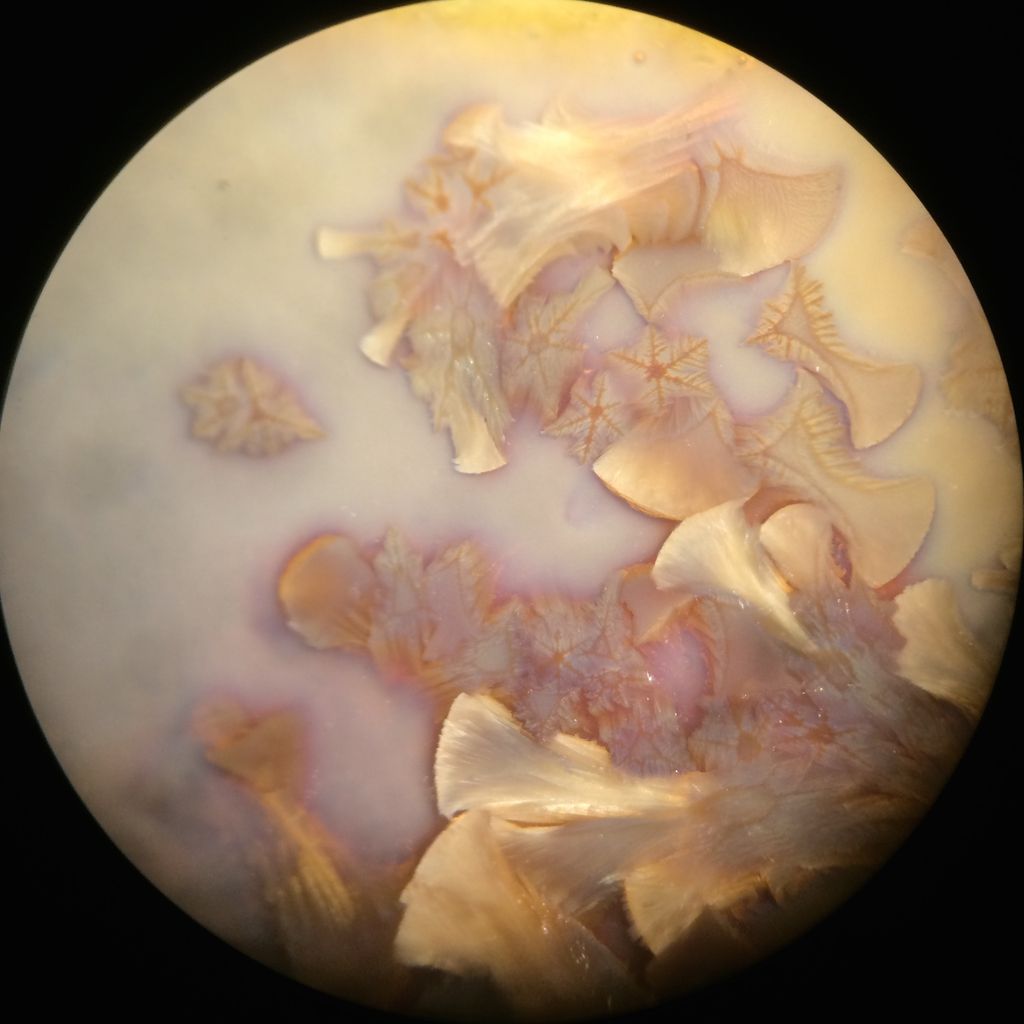 Wow Joerg, those really are fantastic oil spill rainbow fern leaf stars. May I ask what causes the crystals to grow in these shapes? And what size are they? And does Molybdenum give the glaze that blue color or is that from something else? |
|
|
|
Post by tileman2 on Jul 22, 2015 21:16:43 GMT -6
Evan:
Those would be the ones; ty. Gives me something to study.
Tom
Just tried to post a pic: apparently I need to relearn that.
|
|
joerg
New Member
 website: www.crystalsforever.com
website: www.crystalsforever.com
Posts: 49
|
Post by joerg on Jul 24, 2015 14:14:58 GMT -6
Wow Joerg, those really are fantastic oil spill rainbow fern leaf stars. May I ask what causes the crystals to grow in these shapes? And what size are they? And does Molybdenum give the glaze that blue color or is that from something else? Hi, Evan! No idea, what makes them grow like this... there have been elaborate studies with regard to twisted crystals (Bernauer) , too sophisticatedfor me ... :-) Size (those on the foto before) 3 - 5 mm, the others 10 - 20 mm...; the blue comes from cobalt additition. |
|
|
|
Post by evan cornish-keefe on Jul 26, 2015 22:44:20 GMT -6
Tom, the splotchy red photo is just a clear zinc crystalline glaze over red stained slip, the piece was fired 3 times so the red started getting messed up and the crystals grew in a strange way, very three dimensionally on a flat surface. The other image is of a pool of glazes in a dark clay catcher, the runoff of a porcelain piece which looked extremely different with none of the rectangular crystals.
Joerg, I'll take your word on it being too complicated. Do you fire the molybdenum crystal glazes with your zinc crystals, or do they need special treatment? I've wondered if my molybdenum tests should be repeated with a different cooling...?
|
|
joerg
New Member
 website: www.crystalsforever.com
website: www.crystalsforever.com
Posts: 49
|
Post by joerg on Jul 27, 2015 15:22:08 GMT -6
Joerg, I'll take your word on it being too complicated. Do you fire the molybdenum crystal glazes with your zinc crystals, or do they need special treatment? I've wondered if my molybdenum tests should be repeated with a different cooling...? Hi, Evan! how do "personal messages" work here ? - pls send me your email address...  mine is jbaumoeller@gmail.com hasta la vista! J |
|
|
|
Post by tileman2 on Jul 27, 2015 16:22:47 GMT -6
Evan:
The white threads/crystal are of interest to me the most.Tin and some rare earth oxides produce white: but so do salts: potassium and sodium. Usually high amounts of salts take on a milky/ or sometimes pearl hue. You see this in stalagmites in caves which form from alkaline water sources. Sodium and potassium impurities have been on my watch list for study for almost two years now.I ran a series of tests a couple of years back: adding increasing amounts of Custer and G-200 to a base mix with just .25% of CU. I started seeing these white threads, as well as a dramatic increase in spikes and needle/ type crystals. I have a lab type magnifying glass with light: I also put in black light in it at times to studying grazing. I think the pics you and Kuba are posting are fascinating, and gives me a chance to look up close. Very much appreciate both of you doing it.
Tom
|
|
|
|
Post by evan cornish-keefe on Jul 27, 2015 20:59:52 GMT -6
Hey Joerg, it's evanthomas@mac.com , I'll e-mail you Tom, the splotchy red with white crystals is a fairly typical base glaze without coloring oxides, on a red stained cone 6 porcelain. The points on these crystals are at the clay body, growing to the surface in a pool of glaze. The crystals are translucent where thin and get whiter and more opaque as they get thicker. I believe the white is mostly caused by Zinc. I did notice when on tests without alumina that crystals were very transparent and difficult to see, even though they were large. I also tried tin and zircon additions, both caused white glazes maybe more due to masses of crystals than the colorant itself. What rare earth metals are you talking about? Do you have photos of your feldspar tests? Also, here are some white threads, with snowflakes around the edge if you look closely:  |
|
|
|
Post by tileman2 on Jul 31, 2015 15:57:20 GMT -6
Evan:
Just realized that I forgot to answer your questions. Yttrium and Samarium almost always turn white unless they are used in conjunction with other oxides. The colorant strengths of all rare earths (that I have used) are easily overshadowed by standard metal oxides. Neodymium however produces some fun colors. The intensity of colors on a nearly flat surface are different than they are on a vertical surface. Let me dig through some of my old boxes of test tiles: I keep a few of the extreme examples of sodium and potassium poisoning the glaze. High amounts of titanium will likewise discolor to a milky white/greysih area on a flat surface. There are some distinct issues that separate firing horizontal and vertical pieces. On the vertical: 1/4-1/3 and in rare cases 1/2 of the glaze can end up in the catcher. Which also means high percentages of colorants, titanium also end up there. On flat surfaces; it all just sits there and cooks in glaze stew. The green piece you are illustrating is a medium green primarily; with the edges a dark green. Further illustration of how glaze will pool at the lowest point; which means the colorants and titanium is collecting there.
Tom
|
|
|
|
Post by evan cornish-keefe on Aug 2, 2015 7:09:18 GMT -6
Hey Tom,
I'm not sure what you mean by "poisoning the glaze".
I think the formulas I use have a fair bit more alumina than yours, usually 13-26% EPK (.1-.2 Al), they're fluid and change a lot with vertical / horizontal surfaces, but are no where near as fluid as you described.
The green and white piece had 10% CuC added and it was etched in vinegar to bring out the white. It is a very dark clay body, Ti in the glaze turns a muddy brown, so i was surprised to see it turn white.
|
|
|
|
Post by tileman2 on Aug 2, 2015 10:02:01 GMT -6
Evan:
I use no Al at all, and in addition I have removed any additions that have alumina: such as Spodumene or OM4. At one point I was using Spod as a flux and Om4 as a glaze hardener-in part. The TiO2 I cannot measure because it is less than 0.05. Flat surfaces just have some unique parameters of their own.
Tom
|
|
|
|
Post by tileman2 on Aug 5, 2015 12:50:20 GMT -6
Evan:
Poisoning is just a generic term I use for discolorations in the glaze-whitish to light grey. Several causes, excess TiO2, excess colorants, to which rare earth oxides seem to be the biggest offender. Neodymium for sure. Usually not an issue for vertical pieces; not as much anyway.
Tom
|
|
|
|
Post by tileman2 on Aug 16, 2015 22:41:23 GMT -6
Evan:
Not heard anything from you in awhile- any new and interesting pics?
Tom
|
|
|
|
Post by evan cornish-keefe on Aug 18, 2015 8:13:15 GMT -6
Hey Tom,
I've been following but haven't had anything worth posting about. The past month I haven't tested many glazes and have been making pots with some older recipes. I tried a quick test swapping MnO for ZnO, nothing yet. I also quit my job and moved recently, and had been using their microscope at work (while on the clock) to look at my glazes, so now I need to get my own. I've also been thinking of putting a small book together with some of these images.
I'm still not exactly sure what poisoning is referring to, a picture would help, I can think of a few different things that might fit this description. And in your alumina free glazes, do you find the crystals very transparent or difficult to see even if they're large?
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Oct 29, 2015 8:49:15 GMT -6
Hi Evan, You've opened a new and wonderful window into the world of crystals in glazes. After seeing the black snowflake have been looking more closely for microcrystals; recently got this in a 3110 base (willemite) with about 8% TiO2, 2% MnO2 and 1% CoCO3 cone ten. (apologies for the lack of clarity). Gordon  |
|
Kuba
Full Member
   SztukKilka in Old Formu
SztukKilka in Old Formu
Posts: 111
|
Post by Kuba on Nov 7, 2015 3:16:03 GMT -6
|
|