Kuba
Full Member
   SztukKilka in Old Formu
SztukKilka in Old Formu
Posts: 111
|
Post by Kuba on Feb 8, 2017 14:51:34 GMT -6
Hi Bernhard, great knowledge You have. Do You think that during low post fire the glaze is soft enough to crystal structure?
|
|
|
|
Post by Bernhard Schärf on Feb 8, 2017 15:49:54 GMT -6
Hi Bernhard, great knowledge You have. Do You think that during low post fire the glaze is soft enough to crystal structure? Hi Kuba, Crystallization is always possible up to the glass-transition temperature (TG), despite the higher viscosity. The rate of crystallization decreases naturally, of course. Since the nucleation is still large enough, many micro-crystals are formed until finally a matt-glaze is formed. 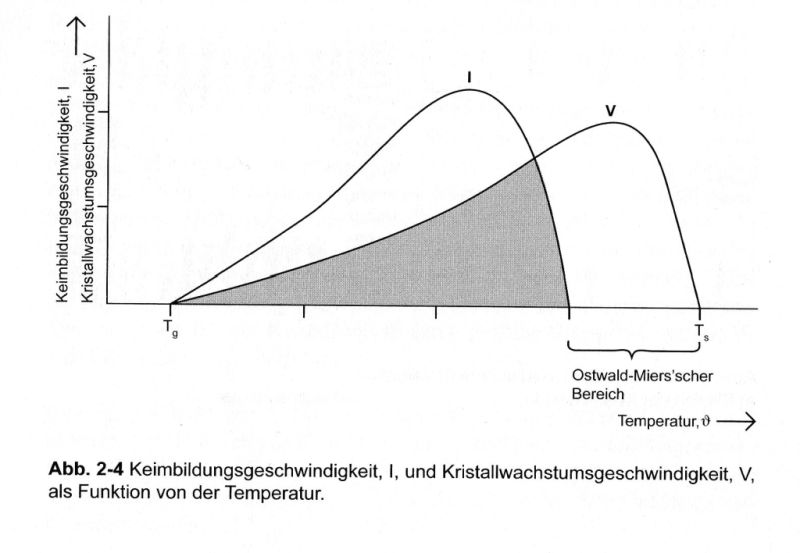 Bernhard |
|
|
|
Post by Arnie Benton on Feb 9, 2017 13:29:43 GMT -6
Here's today's firing - it continues the theme of small changes making a huge difference. 

The first picture is a glaze with Fe Mn Ti
The second picture is the same glaze on the bottom and a second glaze with Cu Co Ti on top.
Here's another interesting result -

This piece has the exact same glazes as in the second piece above, applied in the exact same way, in the same firing, etc - the only difference is that it has the addition of 0.1 gm Fe to the top glaze. I can't really believe that 0.1 gm of Fe could make such a difference! Will repeat this one.
Here's another piece -

It has Ru Cu in the bottom glaze and Cu Co Ti in the top glaze.
And here's a piece with the same pair of glazes, minus the Cu in the bottom glaze -

In this example the difference is 2 gm Cu, so I believe the result - but there is still Cu in the top glaze - so the difference in color of the ground and in the structure of the crystals is amazing to me -
That's enough for me to try to digest in one day.
Arnie
|
|
|
|
Post by Bernhard Schärf on Feb 9, 2017 15:00:39 GMT -6
Here's today's firing - it continues the theme of small changes making a huge difference.
Arnie
----------------------------------------------------------------------------------------------------------
Arni, with such wonderful results one can only answer with Albert Einstein:
"Imagination is more important than knowledge, because knowledge is limited."
Thank you for participating in your development.
I'm currently testing barium-titanium crystal glazes and here, too, I must note that only a few differences in the glaze cause significant changes in color design. Both glazes contain 2% cobalt oxide and in the left glaze is only a higher proportion of titanium oxide.

Bernhard
|
|
|
|
Post by Bernhard Schärf on Feb 10, 2017 2:18:16 GMT -6
Here are the results -
 -------------------------------------------------------------------------------------------------- Arni, I have an idea of trying to test the white splashes : Repeat the above piece with the same glazes, but cool down below 1000 ° C as quickly as possible. If the white ones are still present, then they should be Wollastonite, otherwise they are beta-Willemite and you have to find out which crystal (mirco) it is. In your attempts, you should also monitor the cooling curve with, because after the filling of the furnace, of course, can come to a difference. I wish you success Bernhard |
|
|
|
Post by Arnie Benton on Feb 10, 2017 8:58:16 GMT -6
Hi Bernhard - Here's the piece after the initial crystalline firing - before it was struck -  This is after normal cooling. The final hold is at 1900F so it gets below 1830 pretty quickly - does this answer the question?? Arnie |
|
|
|
Post by Bernhard Schärf on Feb 11, 2017 4:33:40 GMT -6
Here's the piece after the initial crystalline firing - before it was struck -  This is after normal cooling. The final hold is at 1900F so it gets below 1830 pretty quickly - does this answer the question?? ---------------------------------------------------------------------------------------------------------- Yes !!! Here is a test from 2008 to a course with my friend Werner Gnegel at the Kunsthochschule in Kassel. The glaze contains only Cu as color oxide ..... You can see here also white rings and then brown rings. I assume that the white rings at higher temperatures than the brown rings. With corresponding lintel cooling from 950, 900, 850 and 800°C one would certainly recognize more. So, the idea with the wollastonite was a mistake. I assume that the other components Ti, CaCo3 act only as catalysts. Maybe the Cobalt has any influence, because the blue is completely gone in the background. Titanium changes the temperature limit between alpha- and beta-Willemit.  Bernhard |
|
|
|
Post by Arnie Benton on Feb 11, 2017 9:11:46 GMT -6
Bernhard -
So, let's see if I am understanding this - we're talking about heating already glazed pieces to a temperature that causes a change in the structure of the Zinc Silicate crystals - alpha and beta willemite - which will change the colors - maybe the Zinc and Silica still floating in the glaze also form new crystals, combining with the coloring oxides, Ti, etc?
So I just happened to hit upon a combination of ingredients and temperature that produces interesting changes -
Another so - my further experiments can be to make changes in the ingredients, the temperature range - and I'll try just increasing the length of the hold at 1350 F from 1.5 to 3 hours - maybe more time makes a difference also. Does this make sense?
The fact that I'm working with flat surfaces probably is important as well.
Arnie
|
|
|
|
Post by Bernhard Schärf on Feb 11, 2017 12:00:14 GMT -6
Arnie, I'm on the road for a few days, I will refresh my knowledge again and then report more in detail. Bernhard |
|
|
|
Post by Arnie Benton on Feb 17, 2017 12:04:06 GMT -6
I've got 2 ideas swirling around in my brain -
trying to explain, understand what's going on with striking.
Beta willemite is one possibility - crystal structure changes around 1350 degrees F, which results in color changes in ground and crystals.
Curie temperature is another possible factor - temperature at which 'permanent' magnetic forces are lost and the element is influenced by other magnetic forces. Maybe this results in crystal structure changes as with beta willemite. Maybe both of these explanations are describing the same thing?
So I struck some more pieces at 1350 F, took pictures, and re-struck them at 1450 F -
Since the Curie temp for Fe is about 1420 F, I wondered if the Fe in the glaze might be greatly affected - and it was!
But at 1450 F the beta willemite might also change and cause the changes seen.
Here are some examples -


First picture is after striking at 1350 F.
Second picture is the same piece re-struck at 1450 F.
A dramatic change in the Fe color of the ground.
Another example -


Same sequence, first struck at 1350 and then re-struck at 1450. There is no Fe in the glaze. The white color in the ground, thought to be caused by beta willemite, is mostly gone.
So, do we have a different explanation or the same explanation for the 2 sets of pictures.
And what would be a good next step in exploring this phenomenon?
Arnie
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Feb 20, 2017 12:58:28 GMT -6
Hi Arnie, Have you tried a with/without titania comparison? Beta willemite- Mn activates willemite to fluoresce yellow-green under short wave UV. About 2% (no other colorants) will light up the crystals. If you do not have access to short wave UV mail over some test tiles. (will pm address if you wish to do so) I'm skeptical, the experience here is that there is no free willemite in a crystalline glaze and in an experiment adding synthetic fluorescent willemite to the glaze it did not survive . Also its likely that any willemite which does form acts as a nucleus for a crystal. In other words the willemite is all in the crystals formed. Let's see. Gordon  |
|
|
|
Post by Arnie Benton on Feb 20, 2017 15:11:19 GMT -6
Hi Gordon -
I'll try a glaze with no Ti - but my belief at this point is that Ti is necessary to produce the effects from striking that I've been getting.
Also, I'll make some pieces with Mn 2% and see what happens - if they produce the white patches that Bernhard thinks might be Beta willemite, I'll ship them to you -
You don't think that willemite can be produced when a crystalline glaze is re-fired to 13-1400 degrees or so?
Arnie
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Feb 20, 2017 18:16:36 GMT -6
Hi Arnie,
Ok, TiO2 and CaCO3. Are they both necessary? Let's find out.
?"willemite can be produced when a crystalline glaze is re-fired to 13-1400 degrees or so"
It cannot be ruled out but how is such demonstrated? Your test may answer the question right off or not.
Looking forward to your results.
Gordon
|
|
|
|
Post by Bernhard Schärf on Feb 21, 2017 3:46:20 GMT -6
Thanks Arnie for the test .... Super result .... I'm back from my tour today and will answer tomorrow. Bernhard Hello Arnie, I go here completely with Gordon .... Experiments with manganese (Zn2SiO4: Mn) should make the difference between alpha and beta-Willemit, because alpha-Willemit shows green colors and beta-Willemit rather yellowish under UV light. However, manganese will not bring such a color spectrum as copper but as a detection of alpha / beta Willemit is of course well suited. I will buy a UV lamp and report results. I think the combination Cu / Co brings the most extensive color spectrum depending on the coordination number (4 or 6) and the value of the oxides. I believe that the white patches will only occur with Cu. I think that 1450 is still too low to convert all beta-Willemite. Perhaps then, as both kinds are still present, these mixed colors also arise. My thought is 1650 about 30 minutes and all beta-Willemite should be converted into alpha-Willemite. In the experiment without titanium dioxide, as Gordon suggests, the beta-Willemite should be reduced because titania stabilizes the beta phase. The beta-Willemite phase offers several potentially useful proberties. Unlike alpha-Willemite, beta-willemite can have a widely variable composition, ranging from 33 to 67% ZnO. In the crystal-free background, sufficient ZnO might still be present to form new beta-willemit crystals. A Zn(1.7)SiO4 phase is also mentioned in the literature. Bernhard |
|