|
|
Post by Arnie Benton on Jan 25, 2017 13:37:44 GMT -6
Didn't know what to call this thread - I'm still doing what I've been doing - putting one glaze on top of another, firing and then striking - the striking causes dramatic effects in the ground - doesn't happen with all glazes and I'm trying to figure out what glaze ingredients lead to these changes with striking.
When I mix the 2 glazes together and apply as one glaze, the effect doesn't happen.
So the partial mixing of the 2 glazes when applied separately seems to be important -
I've been using a ratio of 1/3 of the lower glaze and 2/3 of the upper glaze. These shallow bowls are 8 3/4 inches in diameter before firing. I'm using a loading of 0.35 gm of dry glaze per square inch, which works out to 21 gms of dry glaze. So the lower glaze has 7 gm of dry glaze and the upper glaze has 14 gm of dry glaze. I mix my dry glazes with water, 1 to 1 by weight so the calculations are easier.
I'm going to pause at this point and make a comment - the details in the above paragraphs make my eyes glaze over, and I know what I'm talking about. For those of you who are relatively new to crystalline glazing it could be enough to get you to try something else - but after 12 years of immersing myself in crystalline glazes I've come to accept that the details are important, no crucial, to have any hope of gaining even a little control of the process.
To continue, in this firing I decided to vary the ratio of top to bottom glaze - so I made it 1/2 of each glaze - the bottom glaze went from 7 to 10.5 gm of the total, and the top glaze from 14 down to 10.5 gm of the total.
Here are the results -
    For each pair the top picture has 1/3 lower glaze and the bottom picture has 1/2 lower glaze. For each pair the top picture has 1/3 lower glaze and the bottom picture has 1/2 lower glaze.
Otherwise each pair is identical.
Also, there is no Fe or Mn in any of the above glazes - so I'm wondering where that ground color that I've been associating with Fe or Mn comes from -
Every experiment I do leads to more questions and after 12 years I'm not bored yet!
Arnie
|
|
Abel
New Member

Posts: 9
|
Post by Abel on Jan 25, 2017 19:47:43 GMT -6
This is very interesting Arnie! You are getting asesome results! I love all the detail that you give about your experimentation process! It is exciting that new approaches continue to be discovered and it is people like yourself that are helping this science to evolve. Keep up the awesome work and please don't ever feel like you are giving too many details. I think that is exactly what we all need. Thanks for sharing this discovery with us! I look forward to hearing more when you feel like sharing again...
Have a great evening!
Abel
|
|
gczop
Full Member
  
Posts: 202
|
Post by gczop on Jan 29, 2017 12:14:21 GMT -6
Arnie,
Thanks for the details. So many tests to do, wish I'd started 30 years ago.
My conjecture is that titanates are formed in the strike process.
In the garage, mixing calcium carb. ,zinc ox., MnO2, lithium carb,etc with titania and putting it in a cone ten firing produces a sintered 'titanate'.
I'm presently experimenting with oilspot glaze with a thin layer of manganese dioxide and titania applied to bisque porcelain under the glaze.
One day may have something presentable to show for all this.
Gordon
|
|
|
|
Post by Arnie Benton on Jan 29, 2017 15:13:14 GMT -6
Abel - For years I approached crystalline glazing as I did stoneware glazing - more of an art than a science. With crystalline I finally got the message - apply the scientific method and only change one variable at a time. That makes for slow progress since we don't see the results until days later.
Arnie
|
|
Kuba
Full Member
   SztukKilka in Old Formu
SztukKilka in Old Formu
Posts: 111
|
Post by Kuba on Feb 4, 2017 13:03:10 GMT -6
The beauty of crystalline glazes is that everyone has his own experience/road and everybody can achieve very different results that become his specialty. I think that You (Arnie) achieved something special that I will always connect with You  |
|
|
|
Post by Arnie Benton on Feb 4, 2017 19:04:10 GMT -6
Thank you, Kuba! This is special for me. To have something so unique that someone will look at it and say - that's by Arnie Benton - is as close to immortality as any of us gets.
|
|
|
|
Post by Bernhard Schärf on Feb 6, 2017 13:23:05 GMT -6
Since the lower glaze has the ingredients - Ti and CaCO3
Arnie
....................................................................................
First my question, what effects do you mean?
The brownish spots or whitish radiating structures?
Since I have been dealing very intensively with Ca-Ti crystal glazes some time ago,
Here some thoughts:
First, the phase diagram of SiO2-TiO2-CaO:
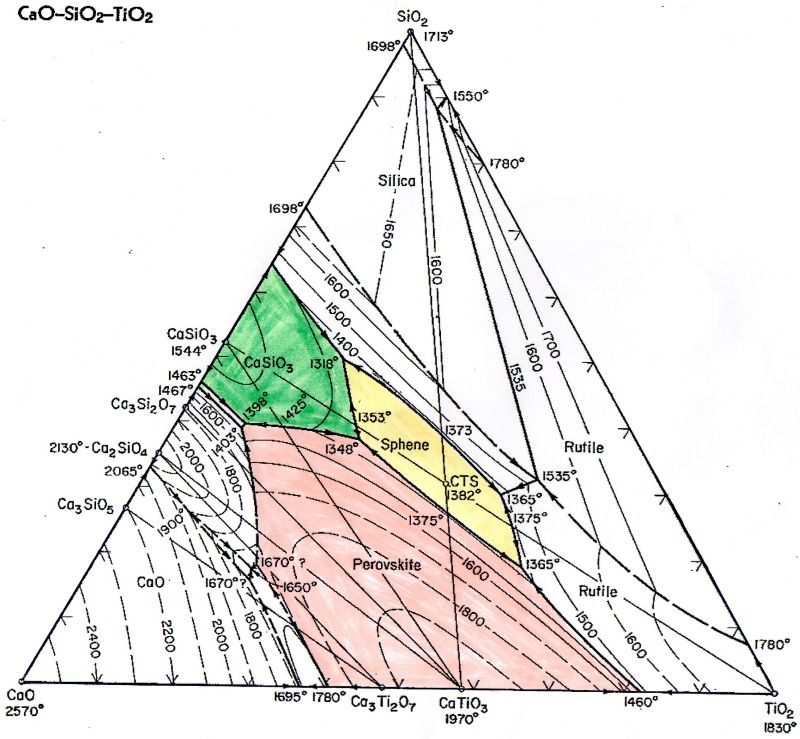 Sphene (CaTiSiO5), perovskite (CaTiO3) and wollastonite (CaSiO3) are formed depending on the composition. I assume in your case that the whitish radiating structures are small wollastonite crystals, because the SiO2 will come from the glaze. In addition, perovskites are formed only at higher temperatures. The TiO2 can then act as nucleating agent and melting point. In my many experiments, the Sphene and Perovskite were always colored, if I used coloring oxides. Since your Willemite also have a bluish coloring, Perovskite or Sphene should also have a coloring. As can be seen from the phase diagram, the green area (wollastonite) is relatively small and since the melting point is strongly dependent on the TiO2 content, one has the exact ratio of CaO, TiO2 and SiO2 to form wollastonite. I myself also had whitish edges in Willemit (Zn2SiO4) crystal glazes with a high proportion of CaO. Here is an example of my Ca-Ti crystal glazes, where I could produce all three crystal species (sphene, perovskite and wollastonite) on a piece by means of a corresponding burning guide.   Hopefully I am not completely wrong and also hope that despite my bad English you can start something with it. Arnie, good luck with your attempts ..... I look forward to further results. Bernhard from Germany ‘the way is the goal’ |
|
|
|
Post by Arnie Benton on Feb 6, 2017 15:15:35 GMT -6
Hi Bernhard -
I'm very happy to see you back on this forum. Not many of the people from 3 to 5 years ago are still posting.
You're diagram shows that I am in the right temperature area - 1300 to 1400 degrees -
Does it make sense that the CaCO3 needs to be near the clay surface and the white flare effect doesn't happen if the CaC03 is in the top glaze and not the bottom glaze? Back up a step - does it make sense that the effect happens only, or mostly, with layered glazes?
I would like to know what you think the brown patches are. They appear even if the glaze has no Iron. The crystals in your picture above are wonderful. I've never seen that growth pattern before.
There are always more questions - and I love that!
Arnie
|
|
|
|
Post by jfox on Feb 6, 2017 17:12:06 GMT -6
|
|
|
|
Post by Bernhard Schärf on Feb 7, 2017 3:25:02 GMT -6
I would like to know what you think the brown patches are. Arnie Thank you Arnie, I'm glad you enjoyed my titanium crystal glazes. For further ideas, I then ask questions about the glaze composition and color oxides. I suspect that the bluish colors are due to nickel oxide (carbonate) As well. Maybe you can say something more about the glass composition. Bernhard |
|
|
|
Post by Arnie Benton on Feb 7, 2017 13:01:48 GMT -6
Hi Bernhard -
The glaze for the first (and second) pictures has Ni Cu in the bottom glaze and Cu Co Ti in the top glaze. In another test I repeated this combination and added more Ni to the bottom glaze, which caused the background to lose the white splashes.
The glaze for the 3rd and 4th pictures has Co Ti in the bottom glaze and Cu Co Ti in the top glaze.
Am I right that these glazes are solid - haven't melted or softened at 1300 to 1400 degrees F? If that is accurate the amount of chemical activity going on within this solid glaze is amazing to me.
Arnie
|
|
|
|
Post by jfox on Feb 7, 2017 21:23:04 GMT -6
|
|
|
|
Post by Arnie Benton on Feb 8, 2017 9:07:02 GMT -6
Hi Jim -
I just read 3 of the articles referenced on this page - so maybe electrical charges are changing in areas of the glaze?
Sure seems like lots of activity does go on within solid structures.
|
|
|
|
Post by jfox on Feb 8, 2017 10:02:37 GMT -6
Hard to say whats actually happening I used to think i understood this stuff but there certainly are alot of intriguing and mysterious processes at work . one example is if you subject a perovskite ,(they primarily use Lead zirconate titanate to a high voltage while it is cooling through the Curie point it becomes polarized, a piezoelectric,meaning the crystal produces a high voltage electric charge if distorted and visa versa. they use them in lighters to produce the spark and many other sensor applications en.wikipedia.org/wiki/Curie_temperature |
|
|
|
Post by Bernhard Schärf on Feb 8, 2017 10:28:00 GMT -6
The glaze for the first (and second) pictures has Ni Cu CaCO3 and Ti in the bottom glaze and Cu Co Ti in the top glaze. In another test I repeated this combination and added more Ni to the bottom glaze, which caused the background to lose the white splashes. The glaze for the 3rd and 4th pictures has Co Ti CaCO3 in the bottom glaze and Cu Co Ti in the top glaze. --------------------------------------------------------------------------------------------- Hello Arni, please decry the late reply, but I was on the way. Thanks for the information on the coloring oxides, so I have some thoughts to the solution. In the old forum I had already written about the forms of the Willemite .... alpha-Willemit and beta-Willemit. Alpha-Willemit is formed above 850-900 ° C and beta-Willemit below. However, with titanium oxide, the temperature range can be raised where beta-Willemit is formed. We know that of Ni-colored Willemites with a high proportion of titanium dioxide, where sometimes unsightly colors are produced. Alpha and Beta-Willemit also have different lattice structures and therefore create different colors (for the same color oxide). The brown spots and the yellowish background can come from nickel or copper (beta-Willemit). My guess is rather the copper. See also .. "The wonders of postfire without the reduction" .... 1350 is the beta-Willemit range .... the alpha-Willemit crystals do not change, but in the background micro-crystals of beta-Willemit arise . A little bit, I wonder, however, that the cobalt has so little influence on the background. Why nickel has influence on the white splashes, maybe the white splashes not a Wollastonit and have something to do with the beta-Willemit .... further thoughts are necessary !!! Here is an example of mine with 2% copper in glaze. The vase was slightly reduced in a second fire, but the brown spots and the brown-yellow background were already before the reduction.   Arnie, good luck finding the nice effects on the narrow degree of their origin and let us continue to participate in the forum here at your results. Thank you, Bernhard |
|

















